Yazılar
FDA Rejects MDMA as PTSD Treatment Due to Insufficient Data
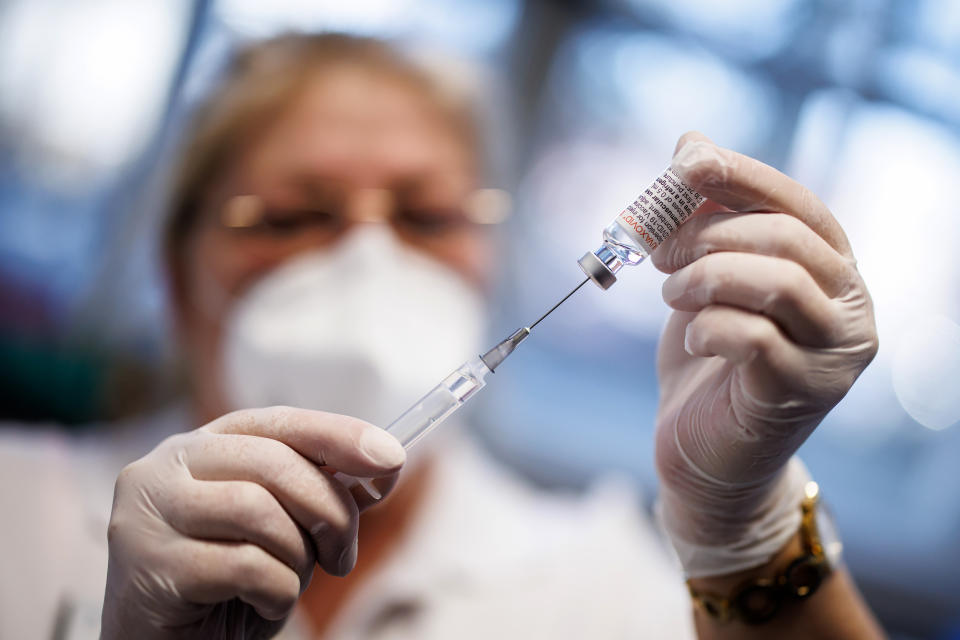
/in Science/tarafından ayaksızThe U.S. Food and Drug Administration (FDA) has denied approval for the use of MDMA, a psychedelic drug commonly known as ecstasy or molly, for treating post-traumatic stress disorder (PTSD). The drug, developed by Lykos Therapeutics, was intended to be used in combination with talk therapy to help individuals suffering from PTSD. However, the FDA raised concerns about the limited clinical data provided, particularly questioning whether psychotherapy contributes to the drug’s effectiveness and whether it is necessary.
The FDA emphasized that the lack of sufficient data prevented them from determining that the drug is both safe and effective for its proposed use. The agency’s decision was influenced by an earlier vote from an expert panel, where the majority expressed doubts about the drug’s overall benefits. In a June panel, 10 out of 11 FDA advisers voted against the effectiveness of MDMA in treating PTSD, citing issues such as the potential lack of long-term benefits, the design of the studies, and the risks associated with heart problems, injury, and abuse.

Lykos Therapeutics expressed disappointment over the FDA’s decision, noting that additional research could take years to complete. The company highlighted the frustration of millions of Americans with PTSD who have seen few new treatment options in over two decades. Despite the setback, Lykos Therapeutics plans to continue working with the FDA to address the agency’s concerns.
This is the first time the FDA has considered a psychedelic drug for medical use. Currently, MDMA remains classified as a Schedule I drug under the Controlled Substances Act, alongside substances like cocaine, heroin, and cannabis, which are defined as having no accepted medical use and a high potential for abuse.
Merck Shares Fall 9% Despite Earnings Beat and Strong Demand for Key Drugs
/in Science/tarafından ayaksızMerck reported second-quarter revenue and adjusted earnings that exceeded Wall Street’s expectations, driven by strong sales from its blockbuster cancer drug Keytruda and other treatments in its oncology and vaccines portfolios, as well as a newly launched cardiovascular drug. Despite this, Merck’s shares fell by 9% due to lighter-than-expected sales of Gardasil, a vaccine for HPV, exacerbated by shipment issues in China.
Merck raised its full-year sales forecast to $63.4 billion to $64.4 billion, slightly up from its previous guidance of $63.1 billion to $64.3 billion. However, it lowered its adjusted profit guidance to a range of $7.94 to $8.04 per share, down from $8.53 to $8.65 per share, reflecting one-time charges for its acquisitions of Harpoon Therapeutics and EyeBio.

For the second quarter, Merck reported adjusted earnings per share of $2.28, surpassing the expected $2.15, and revenue of $16.11 billion, above the anticipated $15.84 billion. The company posted a net income of $5.46 billion, or $2.14 per share, compared to a net loss of $5.98 billion, or $2.35 per share, in the same period last year.
Keytruda recorded $7.27 billion in revenue, up 16% year-over-year, driven by higher uptake for earlier-stage cancers and strong demand for metastatic cancers. Gardasil sales increased by only 1% to $2.48 billion due to shipment timing issues in China. Winrevair, approved in March for treating a progressive lung condition, posted $70 million in revenue, while Capvaxive, a newly approved pneumococcal vaccine, is expected to drive future growth.
Merck’s pharmaceutical division saw a 7% increase in revenue to $14.41 billion. The company’s Type 2 diabetes treatment, Januvia, faced a 27% decline in sales to $629 million due to lower demand, prices, and generic competition. Sales of Merck’s Covid antiviral pill, Lagevrio, fell by 46% to $110 million but still exceeded expectations.
Merck’s animal health division reported $1.48 billion in sales, up 2% from the previous year, but slightly below analyst expectations. Despite strong overall performance, investor concerns about Gardasil sales and future challenges in the pharmaceutical landscape influenced the decline in Merck’s stock.
İlgi çekici linkler
Here are some interesting links for you! Enjoy your stay :)Sayfalar
Arşiv
- Haziran 2026
- Mayıs 2026
- Nisan 2026
- Mart 2026
- Şubat 2026
- Ocak 2026
- Aralık 2025
- Kasım 2025
- Ekim 2025
- Eylül 2025
- Ağustos 2025
- Temmuz 2025
- Haziran 2025
- Mayıs 2025
- Nisan 2025
- Mart 2025
- Şubat 2025
- Ocak 2025
- Aralık 2024
- Kasım 2024
- Ekim 2024
- Eylül 2024
- Ağustos 2024
- Temmuz 2024
- Haziran 2024
- Mayıs 2024
- Nisan 2024
- Mart 2024
- Şubat 2024
- Ocak 2024
- Aralık 2023
- Mayıs 2018
- Şubat 2018
- Aralık 2017
- Kasım 2017