FDA Approves Guardant Health’s Blood Test for Colon Cancer Screening
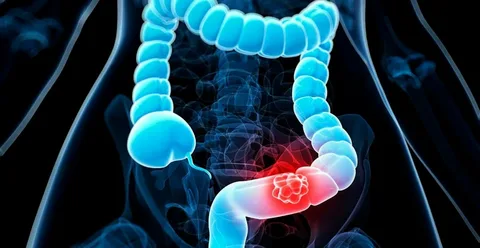
The Food and Drug Administration (FDA) has approved Guardant Health’s blood test, Shield, for colon cancer screening. While not intended to replace colonoscopies, Shield is generating enthusiasm among doctors for its potential to improve screening rates for the second-highest cause of cancer death in the U.S. Previously available at an out-of-pocket cost of $895, Shield’s FDA approval means Medicare and private insurance are likely to cover it, making it more accessible.
Dr. Arvind Dasari from MD Anderson Cancer Center welcomed the approval but noted that the impact on improving screening and reducing mortality remains to be seen. The American Cancer Society estimates over 53,000 deaths from colorectal cancer this year. Research in March showed Shield was 83% effective in detecting colorectal cancers by identifying DNA from cancerous tumors in the bloodstream, though it was less effective at detecting early-stage polyps.
Starting at age 45, Shield would need to be administered every three years. A positive result requires a follow-up colonoscopy to confirm the presence and stage of cancer, as noted by Robert Smith of the American Cancer Society. Shield is the second blood test for colon cancer, following the less accurate and less utilized Epi proColon approved in 2016.
Colorectal cancer rates have risen among those under 55 since the mid-1990s, while rates have declined among those over 60. Dr. William Grady of Fred Hutchinson Cancer Center emphasized that colorectal cancer is preventable with screening, and colonoscopy remains the most accurate method. However, screening rates are low, with fewer than 60% of eligible individuals undergoing recommended screenings. Dr. Sapna Syngal of Dana-Farber Cancer Center highlighted that increasing screening rates through tests like Shield could significantly impact cancer detection.
The March study led by Grady found that people in their 40s and 50s, often busy with work and family, are less likely to undergo screenings. The time-consuming nature and “ick” factor of colonoscopies deter many from the procedure. Electra Paskett of Ohio State University noted that a blood test would be more appealing for many people.
John Gormly, 77, avoided colonoscopies but opted for the Shield test, which led to the detection and successful surgical removal of stage 2 colon cancer. Gormly’s experience underscores the potential life-saving impact of accessible screening options like Shield.